- Reetoo established
- Initial core team formation
- Began development of Gynecology System
- Built a portfolio of five product lines
- GMP factory construction
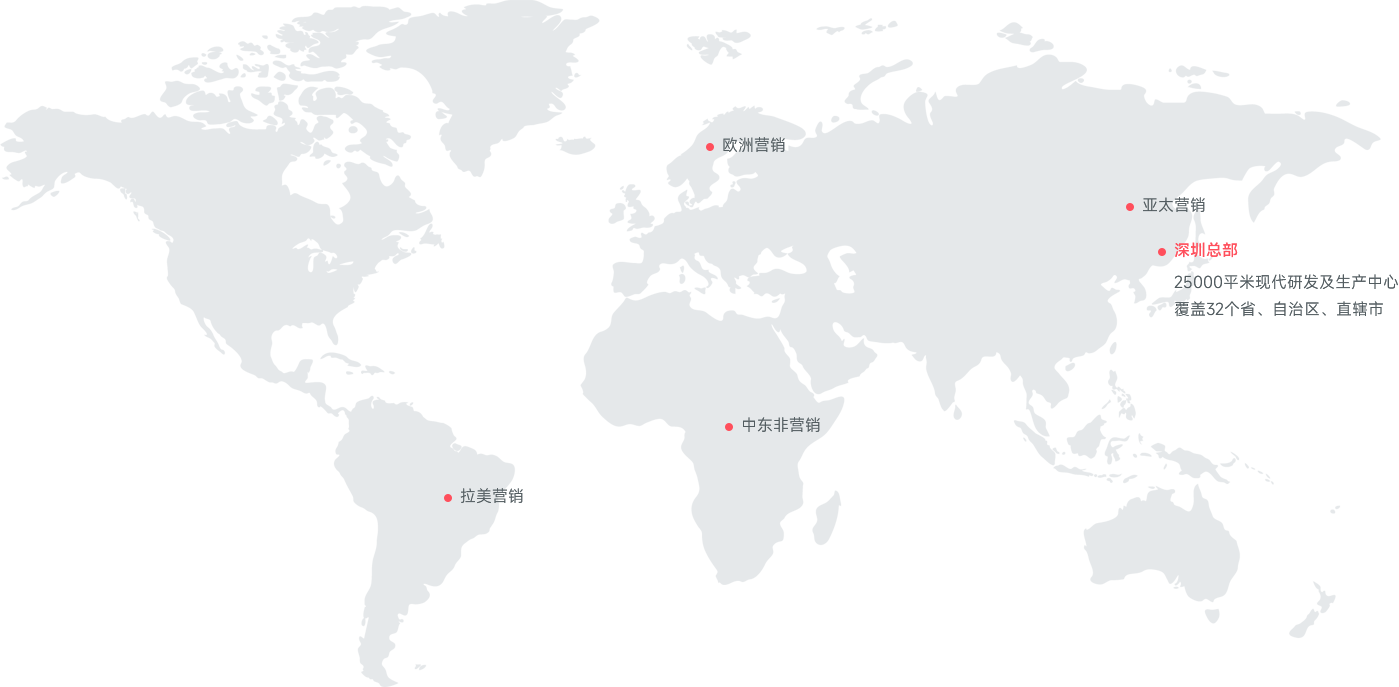
- 深圳总部
Company Established
R&D Staff Proportion
Patents
Trusted by Mid-to-Large Hospitals
Global Installations














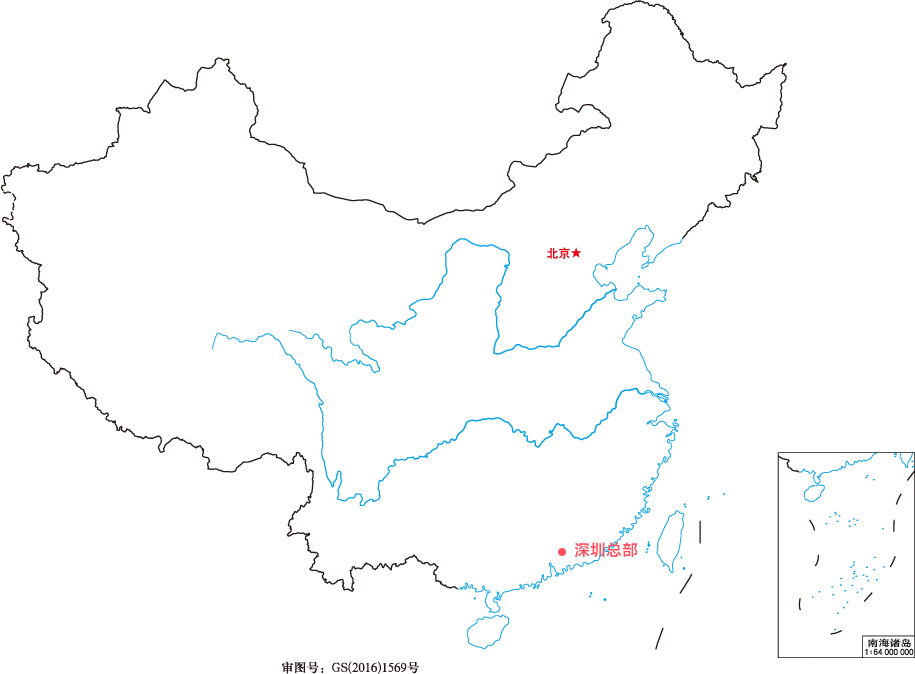
Marketing service centers are located in 32 provinces and cities;
Entered into 1000+ large-scale hospitals in China, including some famous hospitals, like Wuhan Union Hospital of China